
Olive oil is considered to be one of the most healthy dietary fats. There are, however, several types of olive oil present on the market. With the data at present available, the most healthy of them is the virgin olive oil (VOO), rich in phenolic compounds. On November 2011, the European Food Safety Authority (EFSA) released a claim concerning the benefits of the daily ingestion of olive oil rich in phenolic compounds, such as the VOO. In this review, we will summarize the work which have provided scientific evidence of the benefits of VOO consumption over other type of edible oils even olive oils. Data from randomized, controlled human studies, will be presented. This type of studies are able to provide evidence of Level I which is that required for performing nutritional recommendations at population level.
Olive oil is the primary source of fat in the Mediterranean diet which is associated with a low mortality for cardiovascular disease. A large body of knowledge exists providing evidence of the benefits of olive oil consumption on secondary end points for cardiovascular disease. On November 2004, the Federal Drug Administration (FDA) of the U.S.A permitted a claim on olive oil labels concerning: “the benefits on the risk of coronary heart disease of eating about 2 tablespoons (23 grams) of olive oil daily, due to the monounsaturated fat (MUFA) in olive oil” . Recently, results of large EPIC cohorts have reported an inverse relationship between olive oil consumption and coronary heart disease (CHD) mortality and incidence4,5. An inverse relationship between olive oil consumption and stroke risk in women has been reported in the Three-City Study .
Olive oil has major and minor components. The former are the fatty acids, of which the monounsaturated fatty acid (MUFA), the oleic acid, represents from 55% to 83% of the total fatty acids7 .The minor components of olive oil constitute from 1% to 2% of the total content of an olive oil and are classified as: 1) the unsaponifiable fraction, which can be extracted with solvents after the saponi- 40 fication of the oil and contains squalene and other triterpenes, sterols, tocopherol, and pigments, and 2) the soluble fraction that includes the phenolic compounds, the complex forms called polyphenols. Phenolic compounds are present as simple phenols or conjugated forms (Figure 1). Tyrosol (Tyr), hydroxytyrosol (OHTyr), and their secoiridoid derivatives make up around 90% of the total phenolic content of a virgin olive oil. Phenolic compounds from olive oil are the most studied and best known components in terms of their benefits for health. The content of the minor components of an olive oil varies, depending on the cultivar, climate, ripeness of the olives at harvesting, and the processing system employed to produce the olive oil. Olive oils currently present on the market are: extra-virgin, virgin, olive oil (UE, 1991), or pomace (also called orujo) olive oil. Virgin olive oils (VOOs) are those obtained from the fruit of the olive tree solely by mechanical or other physical means under conditions that do not lead to alteration in the oil. Extra-VOOs are VOOs with a free acidity, expressed as g of oleic acid/100 g of olive oil, less than 0.8 g. VOOs with an acidity greater than or equal to 3.3 (International Olive Oil Council Regulation/ T.15/NC.n3.Rev2.Nov24, 2006) or greater than 2 in Europe (European Regulation N. 1513/0) are submitted to a refining process in which some components, mainly phenolic compounds, and to a lesser degree squalene, are lost. By mixing virgin and refined olive oil an ordinary olive oil (olive oil, UE 1991) is produced and marketed. After VOO production, the rest of the olive drupe and seed is submitted to a refining process, resulting in pomace olive oil, to which a certain quantity of VOO is added before marketing.
Here, we will summarize the key work which has provided evidence of the benefits of VOO consumption over other type of edible oils even olive oils. We will focus on data from randomized, controlled human studies, which are those capable to provide the evidence of Level I9 . On the basis of the Evidence-Based Medicine, evidence of Level I is which is required for performing nutritional recommendations at population level.
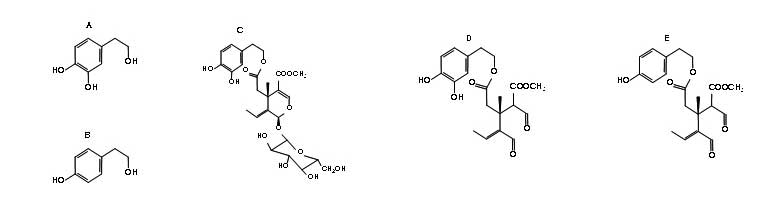

Figure 1: Structures of the major phenolic compounds identified in olives and olive oil. A, hydroxytyrosol; B, tyrosol; C, oleuropein glucoside; D, dialdehydic form of oleuropein (SID-1); E, ligstroside (SID-2); F, aglycone form of oleuropein glucoside (SID-3); G, ligstroside SID-4; H, lignanes (+)-pinoresinol; I: (+)-1-acetoxypinoresinol.
Metabolic disposition of olive oil phenolic compounds in humans from real-life doses of natural olive oils.
One of the prerequisites for assessing the physiological significance of olive oil phenolic compounds is the ability to determine their bioavailability in human beings. Tyr and OHTyr are absorbed by humans from olive oil in a dosedependent manner with the phenolic content of the olive oil administered both after a single dose or after sustained olive oil consumption10,11. This implies an accumulation in the body even from moderate doses of olive oil (25 mL/d) which are lower than the traditional daily dietary intake in Mediterranean countries (30-50g/day). The bioavailability of OHTyr, however, has been shown to change largely depending on the matrix in which the phenolic compounds are administered, the most effective matrix being the olive oil12. Around 98% of Tyr and OHTyr are present in plasma and urine in conjugated forms, glucuronoconjugates or sulfates. This fact indicates the existence of an extensive first-pass intestinal/hepatic metabolism of the ingested primary forms13.
Lipoproteins and Lipoprotein Oxidation
The replacement of saturated fatty acids (SFA) by oils enriched in MUFA versus polyunsaturated fatty acids (PUFA) had similar effects on lowering total and low density lipoproteins (LDL) cholesterol. The PUFA-enriched oil had a slight triglyceride lowering effect, and there was an increase in HDL cholesterol after MUFA consumption in some studies14. There is, however, an issue in which MUFA consumption prevails over the PUFA one: the lipoprotein oxidation. Oleate-rich LDL are less susceptible against oxidation than linoleate-rich particles. This is due to the fact that PUFAs are the key substrate for lipid peroxidation whose propagation chain is going on via the double bonds of the fatty acid15. From 14 studies comparing the resistance of LDL to oxidation only in 2 of them did MUFA-rich diets not promote a higher resistance of LDL to oxidation than PUFA-rich ones16.
Oxidation of the lipid part, or directly of the apolipoprotein (apo) B, of the LDL leads to a change in the lipoprotein conformation by which the LDL is better able to enter into the monocyte/macrophage system of the arterial wall, and develop the atherosclerotic process17. Oxidized LDL has been shown to be independently associated with 10-year coronary artery disease (CAD) events in the general population, and improved the reclassification capacity of Framingham-derived CAD risk functions18.
Phenolic compounds present in VOO have proven to be protective against LDL oxidation. On November 2011, the European Food Safety Authority (EFSA) released a claim concerning the benefits of the daily ingestion of olive oil rich in phenolic compounds, such as the virgin olive oil. The Panel considers that in order to bear the claim, 5 mg of OHTyr and its derivatives (e.g. oleuropein complex and Tyr) in olive oil should be consumed daily. These quantities, if provided by moderate amounts of olive oil, can be easily consumed in the context of a balanced diet19. Here we will revise the main studies which supported this EFSA claim concerning the benefits of phenolic compound-rich olive oil such as VOO.
Human studies on the postprandial effect of olive oil on oxidative stress and oxidative damage
After meals, particularly those rich in fats, a postprandial hyperlipemia and hyperglycemia occurs, which is linked to oxidative stress. Although fasting hyperlipidemia is considered an important risk for cardiovascular disease, postprandial serum lipid levels have been found to correlate more closely to cardiovascular diseases than fasting lipids20. Activation of peroxisome proliferator-activated receptor-α (PPARα) suppresses postprandial lipidemia through fatty acid oxidation in enterocytes21. A functional olive oil enriched with its own phenolic compounds has shown to enhance the gene expression of PPARα in mononuclear cells of pre- and stage I hypertensive patients22.
Several data on the effect of olive oil rich in phenolic compounds, such as VOO, on the postprandial oxidative stress have been reported. They are, however, difficult to compare because some studies do not mention whether or not postprandial lipemia and/or hyperglycemia, which could lead to oxidative stress, occur after olive oil ingestion, while in other studies neither hyperlipemia nor hyperglycemia occur at postprandial state after the olive oil ingestion7, 8 .The ingestion of a 25 mL olive oil dose did not promote postprandial oxidative stress, with independence of the phenolic content of the olive oil, whereas single doses of 40 mL and 50 mL did8. With olive oil doses at which oxidative stress occurs, data from randomized, cross-over, controlled studies in humans showed: i) an increase in the serum antioxidant capacity after VOO ingestion, but not after ordinary olive oil, in comparison with corn oil, suggesting a role for the phenolic compounds of the VOO; and ii) the phenolic content of an olive oil modulates the degree of lipid and LDL oxidation, the lipid oxidative damage being lower after high- than after low-phenolic content olive oil8. In comparison with sunflower oil, meals submitted to a deep frying with olive oils rich in phenolic compounds, both natural and added, have shown to reduce the postprandial oxidative stress in obese people23. Recently, it has been described that food fried in extravirgin olive oil improves postprandial insulin response, an oxidative stress associated phenomenae, in obese, insulin- resistant women24.
Human studies of the sustained effect of olive oil on oxidative stress and oxidative damage
Controversial results have been obtained in short sample size (n<30) human randomized controlled studies performed up to now on the effect of sustained doses of olive oil phenolic compounds on oxidative stress7, 8. Extensive differences existed among the studies in the experimental. design, control of diet, sample population, age of the participants, measurement or not of markers of the compliance of the intervention, as well as in the sensitivity and specificity of the oxidative stress biomarkers evaluated. This fact could explain the differences of results observed among studies.
The results of the EUROLIVE study, however, have provided final evidence of the in vivo protective role of phenolic compounds from olive oil on lipid oxidative damage in humans, at real-life olive oil doses10. The EUROLIVE (The effect of olive oil consumption on oxidative damage in European populations) study was a large, crossover, multicentre, clinical trial performed in 200 individuals from five European countries. Participants were randomly assigned to receive 25 mL/day of three similar olive oils, but with differences in their phenolic content (from 2.7 mg/ kg to 366 mg/kg of olive oil), in intervention periods of 3 weeks preceded by 2 week washout periods. All olive oils increased the HDL-cholesterol and the ratio between the reduced and oxidized forms of glutathione. In the EUROLIVE study, consumption of medium- and high-phenolic content olive oil decreased lipid oxidative damage biomarkers such as plasma oxidized LDL, uninduced conjugated dienes, and hydroxy fatty acids, without changes in F2-isoprostanes. But the most important results of the EUROLIVE study were that the increase in HDL cholesterol and the decrease in the lipid oxidative damage were linear with the phenolic content of the olive oil consumed. The results of the EUROLIVE study provided first level evidence that olive oil is more than a MUFA fat.
Concerning DNA oxidative damage, the urinary excretion of 8-oxo-deoxyguanosine (dGuo) is advocated as a biomarker of the whole body DNA oxidation, a risk factor for cardiovascular and cancer diseases. A protective effect of phenolic compounds from VOO has been reported on in vivo DNA oxidation both in healthy male subjects and in postmenopausal women8. Results of the EUROLIVE study, however, show that consumption of 25 mL of olive oil per day during 3 weeks reduced DNA oxidation in 182 healthy males, as measured by the 24 h urinary excretion of dGuo, irrespective of the olive oil phenolic content25. Differences in the type of population involved (with or without oxidative stress) could explain differences among results. In this sense, one conclusion of the Consensus Report made by the Expert Panel in the International Conference of Olive Oil and Health held in Jaen, Spain, October 20047,26 was that the protective effects on oxidation markers in human trials were better displayed in oxidative stress conditions.
Inflammation, endothelial function, and blood pressure
Other pathological conditions, present in chronic degenerative diseases, such as cardiovascular, cancer, and neurodegenerative, and linked to oxidative stress are inflammation, endothelial dysfunction, hypertension etc. Here we will revise the available information related concerning the effects of VOO consumption on these conditions.
Although the protective mechanism of oleic acid–rich diets on inflammation has been attributed to a decrease in the LDL linoleic acid content, oleic acid is not the single responsible factor for the anti-inflammatory properties of olive oil. Several studies have examined the anti-inflammatory and vasculoprotective effect of olive oil phenolic compounds in humans (Table I). In these studies, VOO with high phenolic content has been shown to be effective in reducing the eicosanoid inflammatory mediators derived from arachidonic acid, such as thromboxane B2 and 6-keto-prostaglandin F1α 27-29, as well as other inflammatory markers, such as high-sensibility C-reactive protein or interleukin- 630,31. A decrease of intracellular adhesion molecule- 1 (ICAM-1) and valscular cell adhesion molecule-1 (VCAM-1) serum levels at postprandial state after VOO when compared with refined olive oil ingestion has been reported32. Also, the postprandial inflammatory response after ingestion of heated oils in obese persons was reduced by the presence of oils with phenolic compounds or non natural antioxidants33. In this study33, VOO or a mix of sunflower and canola oil artificially enriched with olive oil phenolic compounds and other antioxidants, lowered the postprandial nuclear factor ĸappa B (NF-ĸB) activation, increased NF-ĸB inhibitor (IĸB-α), and decreased lipopolysaccharide (LPS) plasma concentration), compared with sunflower oil. In sustained consumption studies, a decrease in ICAM-1 after VOO, but not after VOO plus epigallocatechin-3-galate (EGCG) in early atherosclerosis patients has been reported34. However, no differences in ICAM-1 or VCAM-1 levels were reported after sustained virgin or refined olive oil consumption in coronary heart disease (CHD) patients30.
Vasomotor, haemostatic, antioxidant, and inflammatory activities are involved in endothelial dysfunction, a recent focus of intense research in cardiovascular medicine35. The effect of VOO reducing oxidative stress and oxidative damage, as well as the inflammatory markers has been described above. The postprandial prothrombotic profile improved in healthy subjects and hypercholesterolemic patients after intake of phenol-rich VOO versus a lowphenolic content olive oil2. Several studies have reported beneficial effects of VOO on the endothelial function. An improved post-ischemic hyperemia via reduced oxidative stress and increased nitric oxide metabolites was reported after the intake of phenol-rich VOO in comparison with a low-phenol olive oil in hypercholesterolemic patients36. The effect of VOO phenols on the postprandial endothelial function seems to be mediated by the NOS3 Glu298Asp polymorphism in patients with metabolic syndrome37. Beneficial effects improving the endothelial function have been observed after a 4-month diet with polyphenol-rich olive oil in patients with early atherosclerosis34.
Consumption of olive oil reduces blood pressure2. However, phenolic compounds in VOO play also a beneficial role. A decrease in systolic (SBP) and diastolic blood pressure, with an improvement of endothelial function, has been reported after a 2-month diet with rich-polyphenol olive oil versus a low polyphenol one, in young women with mild hypertension31. In this study31, changes in blood pressure and endothelial function were concomitant with markers related with vasodilatation, such as an increase in nitric oxide and a decrease in serum asymmetric dimethylarginine, as well as a reduction in oxidized LDL and hsCRP. VOO, reduced SBP in hypertensive women and stable CHD patients in comparison with high-oleic sunflower oil and refined olive oil, respectively38. In a parallel study comparing the effect of VOO on blood pressure in diabetic patients and healthy individuals, a reduction in SBP was observed in both conditions39.
Table I: Randomized controlled studies on the effect of VOO on inflammatory and vasculoprotective markers.

TXB2, thromboxane B2; 6-keto- PGF1α, 6-keto-prostaglandin1α; PRP, platelet-rich plasma; LTB4, leukotriene B4, ICAM-1, intercellular adhesion molecule-1; VCAM-1, vascular cell adhesion molecule-1; CHD, coronary heart disease; hsCRP; high sensitivity C reactive protein; IL-6, interleukin 6 ADMA, asymmetric dimethylarginine; TNF-α, tumour necrosis factor alpha; IL-1β, interleukin 1β;MIF, macrophage migration inhibitory factor; JNK, c-Jun Nterminal kinase; LPS, lipopolysaccharide.
Mechanisms by which VOO and its phenolic compounds can exert their benefits in vivo in humans.
Although there is a huge body of experimental studies concerning mechanisms by which VOO and its phenolic compounds could exert their beneficial effects, up-to-date only two have been reported to occur in vivo in humans:
1) an increase of the antioxidant content of the LDL, and
2) a nutrigenomic effect.
Consumption of VOO increases the antioxidant content of the LDL
In human studies the best results obtained on lipid oxidation markers after VOO consumption are displayed in those markers directly associated with LDL. This was one of the conclusions of the Consensus Report made by the Expert Panel in the International Conference of Olive Oil and Health held in Jaen, Spain, October 20047, 26 This could be explained on one hand by the fact that ingestion of any type of olive oil increases the plasma oleic acid content of the LDL lipoprotein. After olive oil ingestion, oleic acid concentration in LDL increased and those of linoleic and arachidonic acid decreased40. As has been referred to before, oleate-rich LDL are less susceptible against oxidation than linoleate-rich particles14. On the other hand, the total polyphenol content bound to human LDL increase in a dose dependent manner with the phenolic content of the olive oil administered10. OHTyr and Tyr metabolites, glucuronides and sulfates, have been reported to bind human LDL after VOO ingestion, but were not detected after refined olive oil ingestion40. An inverse relationship between plasma levels of oxidized LDL and the content of phenols bound to LDL has been reported40. Phenolic compounds which can bind LDL are likely to perform their peroxyl scavenging activity in the arterial intima, where full LDL oxidation occurs17.
Nutrigenomic effect of VOO and its phenolic compounds
Nutrigenomics embrace all “omics” fields, such as genomics, transcriptomics, proteomics, and metabonomics with the aim of understanding and characterizing how nutrients and/or food act at molecular level. Here we will focuse in the transcriptomic data available from human studies, and particularly in those studies in which different types of olive oils have been tested with the same dietary pattern as background. Several studies have examined the transcriptomic profile after VOO, in comparison with low phenolic content olive oil, both at postprandial state and after sustained consumption41. A breakfast based on VOO, high in polyphenols (398 ppm), was able to postprandially repress the expression of proinflammatory genes when compared with a common olive oil based breakfast (low in polyphenols, 70 ppm) in metabolic syndrome individuals. Microarray results showed 19 upregulated and 79 downregulated genes, linked to obesity, dyslipemia, and type 2 diabetes mellitus, after the intake of VOO41. A VOO enriched with its own phenolic compounds enhanced the postprandial expression of cholesterol efflux related genes in vivo in humans versus an standard VOO. In our pre- and stage I hypertensive patients, we observed an increase in ATP binding cassette transporter-A1(ABCA1), scavenger receptor class B type1 (SR-B1), peroxisome proliferator-activated receptors and binding protein (PPARBP, PPARα, PPARγ, PPARδ) and CD36 gene expression in white blood cells at postprandial after high polyphenol VOO when compared with standard VOO22.
Also within the frame of a Mediterranean diet (MeD), in a parallel, controlled, 3-month intervention trial, 90 healthy volunteers were randomized to three intervention dietary patterns: I) MeD supplemented with rich-polyphenol VOO (328mg/kg); II) MeD supplemented with washed VOO (low in polyphenols: 55 mg/kg); and III) habitual diet (control group). Only after the MeD supplemented with rich-polyphenol VOO there was a significant decrease, versus the control group, in the expression of inflammatory genes: interferon gamma (IFNγ), Rho GTPase activating protein 15 (ARHGAP15), and interleukin 7 receptor (IL7R); and oxidative stress: adrenergic b-2 receptor (ADRB2). Changes in gene expression after the MeD supplemented with VOO were concomitant with decreases in lipid oxidative damage and systemic inflammation markers42. In the frame of the PREDIMED study, two studies have reported differences when an enrichment of the MeD with VOO occurs. In the first43, a 3-month intervention with VOO-enriched MeD prevented the increase in cyclooxigenase-2 (COX2) and LDL receptor-related protein (LRP1) genes, as well as reduced the expression of monocyte chemoattractant protein (MCP1) gene, compared with a MeD enriched with nuts or with a low-fat diet. In the second, study44, we compared 3-month changes in the whole genome of peripheral blood mononuclear cells. Results of functional annotation analyses showed that from 18 cardiovascular canonical pathways, 9 were modulated by a MeD enriched with VOO and 4 when the MeD was enriched with nuts44. In a sub-study of the EUROLIVE study, we proposed for the first time an integrated scheme for the in vivo downregulation of the CD40/CD40L system and its downstream products promoted by the consumption of VOO45. Our results showed a decrease in the expression of pro-atherogenic genes: CD40 antigen ligand (CD40L), interleukin 23A (IL23A), ADRB2, oxidized LDL (lectin-like) receptor 1 (OLR1), interleukin-8 receptor-a (IL8RA), and IL7R) after consumption of VOO high in polyphenols when compared with the refined olive oil, low in polyphenols. The decrease in these genes was concomitant with the decreasing trend of interlinked ones such as vascular endothelial growth factor (VEGF), ICAM-1, and MCP1. In this study45, the reduction in LDL oxidation and the increase in antioxidant polyphenols in LDL promoted by the regular dietary intake of phenolic compound-rich VOO, were associated with a down regulation in the expression of genes related with the CD40/CD40L pathway. Due to all referred above, VOO and its phenolic compounds can modulate the expression of atherosclerosis related genes towards a protective mode in vivo in humans.
Conclusions
We have scientific data providing level I evidence that VOO can promote additional benefits to those of provided by other vegetable oils on risk factors for cardiovascular disease such as lipids and lipid oxidation, inflammation, endothelial dysfunction and systolic blood pressure. At present, the known mechanisms by which VOO can exert these beneficial effects in humans arethe increase in the antioxidant content of LDL, and a nutrigenomic effect, modulating towards a protective mode the expression of atherosclerosis-related genes.
Acknowledgements
CIBEROBN is an initiative of the Instituto de Salud Carlos III, Madrid, Spain.